The NICE type 2 diabetes guideline update: taking a cardiorenal metabolic approach
Beverley Bostock RN MSc MA QN, President-Elect Primary Care Cardiovascular Society, ANP Long-Term Conditions Mann Cottage Surgery, Moreton-in-the-Marsh
Practice Nurse 2026;56(3):11-15
The recently updated guidance on type 2 diabetes takes account of current evidence and product licences and aligns much more closely with international guidelines. All clinicians should be aware of the recommendations, even if they are not prescribers
Type 2 diabetes (T2D) remains a major public health challenge, as the prevalence continues to increase along with the associated risk of complications.Diabetes UK states that over 4 million people in the United Kingdom are living with a diagnosis of T2D and that a further 1.3 million have T2D but remain undiagnosed.1 Furthermore, the charity estimates that 6.3 million people are at an increased risk of type 2 diabetes in the UK based on records of blood sugar levels. This mean around 12 million adults in the UK are living with diabetes or prediabetes.
The majority of the cost of treating diabetes (60%, in fact) goes on the management of complications, including microvascular disease such as nephropathy, neuropathy and retinopathy, and macrovascular disease, such as heart attacks, strokes and peripheral arterial disease.2This presents a major challenge to the NHS as well as being a cause of a high level of morbidity and mortality to the individuals living with the condition.In February 2026, NICE published an important update to its guidelines for the management of T2D.3 This update has, for the most part, been welcomed by the diabetes healthcare professional community as it recognises the importance of tailoring care to the individual, with the aim of reducing cardiorenal complications.The guidance has been updated by taking account of current evidence and product licences and aligns much more closely with international guidelines, such as those published by the American Diabetes Association (ADA) and the European Society for the Study of Diabetes (EASD).4The previous NICE guidelines were considerably out of date, specifically when it came to positioning the GLP-1 RA class of drugs such as semaglutide and tirzepatide. This failure was too often replicated in local formularies which continued to relegate these drugs to a fourth-line option, ignoring the potential benefits of semaglutide and tirzepatide for a host of people with T2D. This new guideline will therefore support healthcare professionals to deliver high-quality, evidence-based care. All clinicians should be aware of the recommendations, even if they are not prescribers, as it is important to understand which drugs are recommended for cardiorenal protection so that patients can make informed choices. They can then be directed to a prescriber who can support them to choose medication using a shared decision-making approach. This article includes an overview of the key changes in NG28 and uses a case study-based approach to demonstrate the practical application of the new guideline.
WHAT IS NEW IN THE UPDATE?
The 2026 update revisits the place and evidence for a broad range of glucose-lowering therapies, incorporating evidence from clinical trials, and highlighting how treatment tailored to the individual is essential for reducing future complication risk. The previous NICE update to the T2D guidelines included a focus on the early introduction of the sodium-glucose cotransporter 2 inhibitors (SGLT2i) such as canagliflozin, dapagliflozin and empagliflozin because of the increasing evidence for their cardiorenal benefits.5 However, another class of medication, the glucagon-like peptide-1 receptor agonists (GLP-1 RAs), were not included in that update despite the substantial body of evidence for their positive impact on glycaemic control, weight loss and cardiovascular outcomes.6 This meant that the recommendations for this class of drugs were significantly out of date and did not reflect the evidence for their use in people living with T2D and obesity or atherosclerotic cardiovascular disease (ASCVD).
It is important to note that NICE continues to emphasise lifestyle changes as the foundation of diabetes management. Reference is made to the diabetes remission programme as being something that should be considered when discussing options with people living with T2D: https://www.england.nhs.uk/diabetes/treatment-care/diabetes-remission/. All patients should receive education about diabetes and its potential impact on lifestyle using culturally appropriate materials and support.NICE provides a link to the Better Health resource for more information: https://www.nhs.uk/better-health/
However, the most important changes in this update refer to the use of appropriate pharmacological management, with decisions about which treatment to use based on the existence or risk of complications. NICE recommends taking into consideration any pre-existing cardiovascular disease (CVD), and in the absence of this, undertaking an assessment of cardiovascular risk in order to personalise treatment options. Cardiovascular risk assessment tools such as QRisk 3 or QRisk Lifetime can be used to identify those at increased risk of complications. Along with cardiovascular status, renal function, frailty, overweight and obesity, and heart failure should be taken into account, and consideration should be given to whether the individual has early-onset T2D, defined as occurring before the age of 40 years.
FIRST-LINE THERAPY – METFORMIN, WITH A TWIST
Metformin remains the first-line treatment for people with T2D, unless contraindicated, because of its role in improving insulin resistance and reducing gluconeogenesis. However, the new guidance recommends the use of modified release metformin (MRM) as standard, whereas the previous guidelines only recommended this for people who had tried standard-release tablets and were unable to tolerate the gastrointestinal side effects. Modified release formulations improve tolerability and adherence, allowing the maximum dose to be prescribed in order to optimise glycaemic control. This could result in a lower tablet burden too, as MRM can be prescribed in 1g doses, with the patient only needing to take them once a day, whether they are taking 1g or 2g daily.However, the combination tablets of metformin and an SGLT2 inhibitor only come with standard release metformin so this may hinder the use of these combinations. It will be interesting to see if local prescribing guidelines endorse the use of MRM, as historically it was more expensive than standard-release metformin, although NICE points out that it is currently cheaper.
SGLT-2 INHIBITORS – FOR (ALMOST) ALL
The SGLT-2 inhibitors canagliflozin, dapagliflozin and empagliflozin are now recommended for all people with T2D, unless contraindicated (and contraindications are few). The rationale for this is that they offer cardiorenal protection and people with T2D are at high risk of these complications.7 As previously noted, most of the money spent on T2D is used to treat the complications of the condition, so increasing the use of drugs which offer additional protection to the heart and kidneys can be seen as an investment in individuals, society and the NHS. SGLT-2 inhibitors also support weight loss, which is important in T2D as most people with T2D are living with overweight and obesity.8 The SGLT2i can be introduced as soon as the patient is on the maximum tolerated dose of metformin.
Throughout the guideline it is recommended that any medication is uptitrated to the maximum tolerated dose before additional drugs are introduced. This does not mean that clinicians need to wait three months after initiating metformin to review the HbA1c before adding in an SGLT2i, as the use of these drugs is for cardiorenal protection, whether or not the HbA1c is at target. It should be possible to start someone on 1g of MRM, uptitrate to 2g in a matter of weeks and then add in the SGLT2i. After three months on both drugs, the HbA1c can be measured and further glucose-lowering medication added if needed.
For those unable to take MRM, SGLT2 inhibitors may be a suitable alternative. Contraindications to SGLT2i include being on dialysis or having a previous history of diabetic ketoacidosis (DKA), a complication which is more commonly seen in type 1 diabetes, but which has very occasionally been seen in euglycaemic T2D.9 Caution should be used in people at risk of volume depletion (dehydration) and patients should be reminded that they need to drink more when they are taking this class of medication.
NICE reminds clinicians to teach patients to follow the sick day rules, pausing their use of SGLT2i, ACE inhibitors, metformin, angiotensin receptor blockers and non-steroidal anti-inflammatory drugs (which should ideally be avoided in diabetes anyway) during any period of acute illness and restarting their medication once they are well again.
The Care Quality Commission advises practices to clearly document the advice they have given to people prescribed SGLT2i, although the evidence suggests that for the majority of people, the advantages far outweigh the risks.This is why NICE is keen to promote their use in a greater number of people living with T2D.Of note, in trials of the SGLT2i drugs and in real world studies, Fournier’s Gangrene was at least as common, and sometimes more common in people not taking SGLT2i drugs as in those taking them.10-12 Although it is medicolegally important to inform people of the potential side effects of any medication, it is morally and ethically essential to put side effects into perspective. It is currently estimated that only around 20% of people who qualify for an SGLT2i are actually prescribed one, so there is clearly work to be done and NICE highlights this health inequality as being one which must be rectified.13
BEYOND THE M2 (metformin + SGLT2i)
It is pleasing to see that NICE has developed different approaches to diabetes management based on key patient profiles, but there are also some glaring omissions which may challenge the clinician who is keen to tailor treatments beyond the NICE advice.The categories NICE has identified are people living with T2D and:
- No relevant complications
- Obesity
- Chronic kidney disease (CKD)
- Heart failure (any ejection fraction)
- Atherosclerotic cardiovascular disease (ASCVD)
- Frailty, and
- People with early-onset T2D
As a reminder, almost all of these patients should now be offered both MRM and an SGLT2i, the possible exception being those living with frailty.
With metformin and an SGLT2i (the ‘M2’ approach) being recommended for most people with T2D, decisions will need to be made about if and when to move to a third drug and to determine what that drug might be. The ADA/EASD guidelines recommend taking the M21 – i.e., metformin, an SGLT2i and a GLP-1 RA – approach to reduce the risk of complications in T2D.4 This is where the guidelines diverge. Although NICE has recognised that the evidence for GLP-1 RAs has increased exponentially since their initial guidance, they have only reflected some of that evidence in this update.
WHERE NOW FOR THE GLP-1 RAs?
An important new indication for subcutaneous semaglutide (1mg) is as a standard treatment alongside MRM and an SGLT2i for people living with T2D and ASCVD. While this is to be celebrated, it is disappointing that NICE seems to endorse the use of GLP-1 RAs or tirzepatide in people living with T2D and obesity only for those who have not reached their glycaemic target. The impact of obesity on insulin resistance, glycaemia, blood pressure, lipids and inflammatory status is well-documented, and the omission of these drugs as an option to treat obesity in T2D is a missed opportunity and one which is difficult to comprehend or justify, from a clinical perspective. Of course, NICE is charged with the responsibility of determining the cost implications of any guidance, which may explain this decision, but it does feel short-sighted.
In the ADA/EASD treatment algorithm, the drugs which are deemed to have the greatest impact on both glycaemic control and weight are tirzepatide and semaglutide.4 If clinicians are using tirzepatide or semaglutide in T2D primarily for weight loss benefits, they are reminded to refer to the NICE guidelines on obesity management, which will limit their use of these important drugs.14
In the ADA/EASD guidelines section on the use of insulin, clinicians are advised to consider a GLP-1 RA before insulin, or after a basal insulin has been initiated but before mealtime doses of insulin are introduced.4 This has not been reflected in the NICE update.There is a section from NICE on insulin-based therapies which states that insulin should only be initiated by someone trained in this area.3
FURTHER TREATMENT OPTIONS
With respect to other treatment options, the DPP4 inhibitors (sitagliptin and linagliptin) can be an option for people unable to tolerate other drugs, or as a third line option for people already on two other classes of drugs who have not reached their target HbA1c. They are also recommended for people living with frailty as they are generally well tolerated, with a negligible risk of hypoglycaemia.However, they have no impact (positive or negative) on cardiorenal outcomes or weight. In the ADA/EASD guidelines, they are described as having an intermediate impact on glycaemic levels, which is the lowest category of glycaemic effect.4 Sulfonylureas (SUs), pioglitazone and insulin are generally reserved for cases where multiple medications are needed to reach the target HbA1c and where contraindications limit other options. However, it should be remembered that SUs have no cardiorenal benefits (and may be associated with harm), are associated with weight gain and increase the risk of hypoglycaemia, meaning that home blood glucose monitoring will be required. Pioglitazone is associated with an increased risk of fractures and is contraindicated in heart failure, so it was odd to see that the guidelines seem to advise caution regarding the use of SGLT2i in frailty, because of the risk of hypotension and volume depletion, but recommend considering an SU, pioglitazone or insulin instead. All of these drugs have the potential for significant side effects, especially in frailty, so NICE does include a caveat that SUs and insulin increase the risk of hypoglycaemia and falls. People living with frailty are also more likely to have CKD or heart failure, and NICE, along with the ADA/EASD guidelines, recommends the use of SGLT2i in both of these conditions based on the clear evidence of benefit.3,4Clinicians should therefore carefully consider the risk: benefit profile of all of these medications with each individual.For help with assessing frailty, NICE provides a link to the relevant section in the multimorbidity guidelines: https://www.nice.org.uk/guidance/ng56/chapter/Recommendations#how-to-assess-frailty
OTHER KEY MESSAGES
Other key messages which should be noted from this update include:
- Consider alternative diagnoses if there is a limited response to treatment
- As a reminder, almost all of these patients should now be offered both MRM and an SGLT2i, the possible exception being those living with frailty.
- With metformin and an SGLT2i (the ‘M2’ approach) being recommended for most people with T2D, decisions will need to be made about if and when to move to a third drug and to determine what that drug might be.
The ADA/EASD guidelines recommend taking the M21 approach to reduce the risk of complications in T2D – i.e., metformin, an SGLT2i and a GLP-1 RA.4 This is where the guidelines diverge. Although NICE has recognised that the evidence for GLP-1 RAs has increased exponentially since their initial guidance, they have only reflected some of that evidence in this update.
WHERE NOW FOR THE GLP-1 RAs?
An important new indication for subcutaneous semaglutide (1mg) is as a standard treatment alongside MRM and an SGLT2i for people living with T2D and ASCVD. While this is to be celebrated, it is disappointing that NICE seems to endorse the use of GLP-1 RAs or tirzepatide in people living with T2D and obesity only for those who have not reached their glycaemic target. The impact of obesity on insulin resistance, glycaemia, blood pressure, lipids and inflammatory status is well-documented, and the omission of these drugs as an option to treat obesity in T2D is a missed opportunity and one which is difficult to comprehend or justify, from a clinical perspective. Of course, NICE is charged with the responsibility of determining the cost implications of any guidance, which may explain this decision, but it does feel short-sighted.
In the ADA/EASD treatment algorithm, the drugs which are deemed to have the greatest impact on both glycaemic control and weight are tirzepatide and semaglutide.4 If clinicians are using tirzepatide or semaglutide in T2D primarily for weight loss benefits, they are reminded to refer to the NICE guidelines on obesity management, which will limit their use of these important drugs.14
In the ADA/EASD guidelines section on the use of insulin, clinicians are advised to consider a GLP-1 RA before insulin, or after a basal insulin has been initiated but before mealtime doses of insulin are introduced.4 This has not been reflected in the NICE update. There is a section from NICE on insulin-based therapies which states that insulin should only be initiated by someone trained in this area.3
FURTHER TREATMENT OPTIONS
With respect to other treatment options, the DPP4 inhibitors (sitagliptin and linagliptin) can be an option for people unable to tolerate other drugs, or as a third line option for people already on two other classes of drugs who have not reached their target HbA1c. They are also recommended for people living with frailty as they are generally well tolerated, with a negligible risk of hypoglycaemia. However, they have no impact (positive or negative) on cardiorenal outcomes or weight. In the ADA/EASD guidelines, they are described as having an intermediate impact on glycaemic levels, which is the lowest category of glycaemic effect.4 Sulfonylureas (SUs), pioglitazone and insulin are generally reserved for cases where multiple medications are needed to reach the target HbA1c and where contraindications limit other options. However, it should be remembered that SUs have no cardiorenal benefits (and may be associated with harm), are associated with weight gain and increase the risk of hypoglycaemia, meaning that home blood glucose monitoring will be required. Pioglitazone is associated with an increased risk of fractures and is contraindicated in heart failure, so it was odd to see that the guidelines seem to advise caution regarding the use of SGLT2i in frailty, because of the risk of hypotension and volume depletion, but recommend considering an SU, pioglitazone or insulin instead. All of these drugs have the potential for significant side effects, especially in frailty, so NICE does include a caveat that SUs and insulin increase the risk of hypoglycaemia and falls. People living with frailty are also more likely to have CKD or heart failure, and NICE, along with the ADA/EASD guidelines, recommends the use of SGLT2i in both of these conditions based on the clear evidence of benefit.3,4 Clinicians should therefore carefully consider the risk: benefit profile of all of these medications with each individual. For help with assessing frailty, NICE provides a link to the relevant section in the multimorbidity guidelines: https://www.nice.org.uk/guidance/ng56/chapter/Recommendations#how-to-assess-frailty
OTHER KEY MESSAGES
Other key messages which should be noted from this update include:
- Do not switch from standard release metformin to MRM if people are tolerating the former well
- Continue medications which have helped the patient to reach HbA1c or weight goals even after these goals have been achieved
- Continue SGLT2i drugs for cardiorenal protection, even if the HbA1c target has not been met
- Stop the GLP-1 RAs drugs or tirzepatide if the BMI is below 18.5kg/m2
- Stop the GLP-1 RAs drugs or tirzepatide if there has been no improvement in the HbA1c and the drugs are not being given for cardioprotection
- Add semaglutide 1mg for anyone who develops ASCVD after their T2D diagnosis
THE DIABETES CLINIC
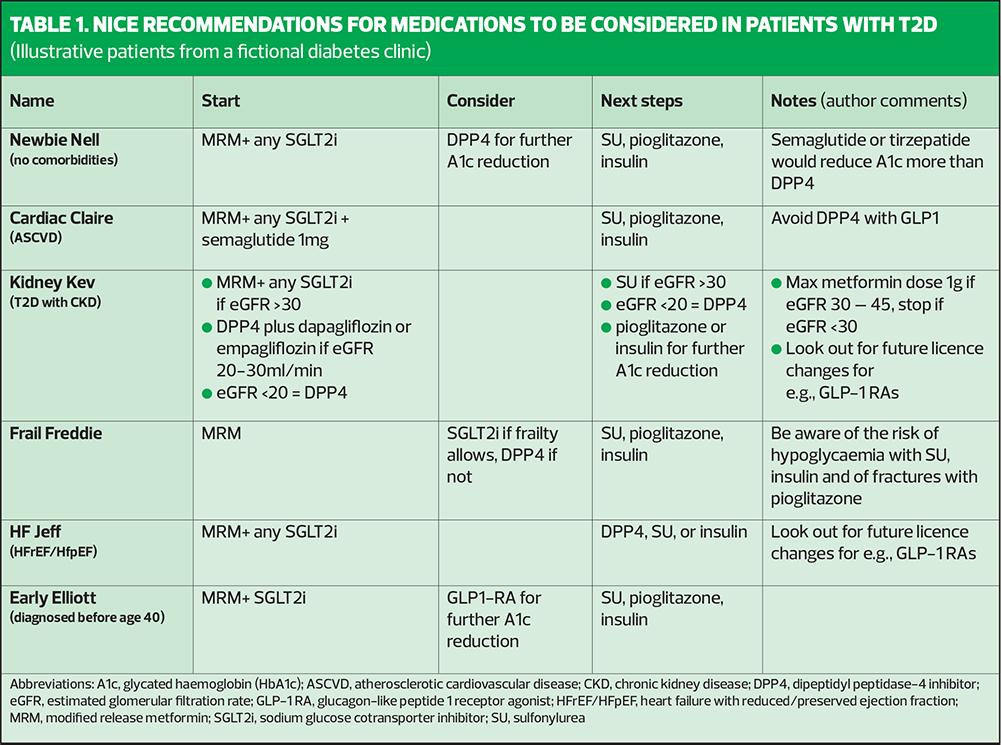
Implementing new guidelines can be challenging, especially with the different categories included in the update and the variations between NICE advice and that from the ADA/EASD guidelines. As a result, consider the people in our fictional T2D clinic: Newbie Nell, with no comorbidities, Cardiac Claire with ASCVD, Overweight Olive with a BMI of 30+, Kidney Kev with chronic kidney disease, frail Freddie, HF Jeff with heart failure, and Early Elliot who developed T2D before he was 40 years old.
The updated recommendations for treatments are summarised in Table 1, which uses the fictional cases described above to illustrate the relevant medications that should be considered.
The focus here is on the drugs discussed in the new guidelines but it is essential to remember that the management of blood pressure and lipids is a key component of optimising outcomes in T2D.
- Do not switch from standard release metformin to MRM if people are tolerating the former well
- Continue medications which have helped the patient to reach HbA1c or weight goals even after these goals have been achieved
- Continue SGLT2i drugs for cardiorenal protection, even if the HbA1c target has not been met
- Stop the GLP-1 RAs drugs or tirzepatide if the BMI is below 18.5kg/m2
- Stop the GLP-1 RAs drugs or tirzepatide if there has been no improvement in the HbA1c and the drugs are not being given for cardioprotection
- Add semaglutide 1mg for anyone who develops ASCVD after their T2D diagnosis
IN SUMMARY
The NICE T2D guidelines have been updated to reflect the increasing body of evidence for preventing and managing the cardiorenal complications of this condition. This is an important development which underlines the link between obesity, T2D, CKD and CVD. Clinicians should be aware of this update and should support people living with T2D to understand why extra medications are being recommended to protect them from future life-limiting complications. It is also important to recognise that the evidence for the management of cardiorenal metabolic disease is moving on at a great pace and that implementing evidence, and not just guidelines, is at the heart of best practice.
References
- Diabetes UK. How many people in the UK have diabetes? 2025. https://www.diabetes.org.uk/about-us/about-the-charity/our-strategy/statistics
- Hex N, MacDonald R, Pocock J, et al. Estimation of the direct health and indirect societal costs of diabetes in the UK using a cost of illness model. Diabetic Med 2024;4(9):e15326. https://doi.org/10.1111/dme.15326
- NICE NG28. Type 2 diabetes in adults: management; 2026 https://www.nice.org.uk/guidance/NG28
- Davies MJ, Aroda VR, Collins BS, et al. Management of Hyperglycemia in Type 2 Diabetes, 2022. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes care 2022;45(11):2753–2786. https://doi.org/10.2337/dci22-0034
- Vallon V. State-of-the-Art-Review: Mechanisms of Action of SGLT2 Inhibitors and Clinical Implications. Am J Hyperten 2024;37(11):841–852. https://doi.org/10.1093/ajh/hpae092
- Liu QK. Mechanisms of action and therapeutic applications of GLP-1 and dual GIP/GLP-1 receptor agonists. Front Endocrinol 2024;15:1431292. https://doi.org/10.3389/fendo.2024.1431292
- Yun JS, Ko SH. Current trends in epidemiology of cardiovascular disease and cardiovascular risk management in type 2 diabetes. Metabol Clin Experiment 2021;123:154838. https://doi.org/10.1016/j.metabol.2021.154838
- Preda A, Montecucco F, Carbone F, et al. SGLT2 inhibitors: from glucose-lowering to cardiovascular benefits. Cardiovasc Res 2024;120(5):443–460. https://doi.org/10.1093/cvr/cvae047
- Morace C, Lorello G, Bellone F, et al. Ketoacidosis and SGLT2 Inhibitors: A Narrative Review. Metabolites 2024;14(5):264. https://doi.org/10.3390/metabo14050264
- Heerspink HJL, Stefánsson BV, Correa-Rotter R, et al & DAPA-CKD Trial Committees and Investigators. Dapagliflozin in Patients with Chronic Kidney Disease. New Engl J Med 2020;383(15):1436–1446. https://doi.org/10.1056/NEJMoa2024816
- Yang JY, Wang T, Pate V, et al. Real-world evidence on sodium-glucose cotransporter-2 inhibitor use and risk of Fournier's gangrene. BMJ Open Diabet Res Care 2020;8(1):e000985. https://doi.org/10.1136/bmjdrc-2019-000985
- Petruski-Ivleva N, Schneeweiss S, Eapen S, et al. Fournier's gangrene in patients with type 2 diabetes using second-line antidiabetic medications. Diabet Obes Metabol 2020;22(2), 267–271. https://doi.org/10.1111/dom.13886
- Ozaki AF, Ko DT, Chong A, et al. Prescribing patterns and factors associated with sodium-glucose cotransporter-2 inhibitor prescribing in patients with diabetes mellitus and atherosclerotic cardiovascular disease. CMAJ Open 2023;11(3):E494–E503. https://doi.org/10.9778/cmajo.20220039
- NICE NG246. Overweight and obesity management; 2026. https://www.nice.org.uk/guidance/ng246
Related articles
View all Articles